What is spectroscopy?
Spectroscopy is the name given to one of the studies in science. In the past, spectroscopy was a term for the study of the interaction between radiation and matter as wavelength ('λ') – for example, a prism.
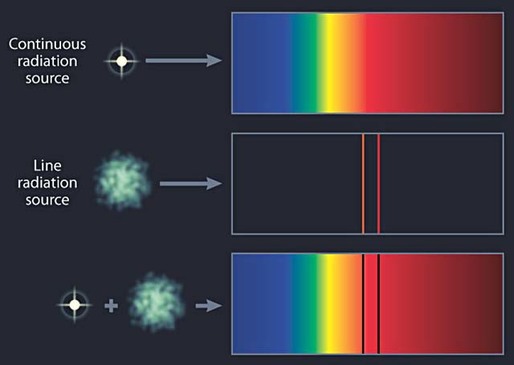
Today, spectroscopy is essentially a scientific measurement technique. It measures light that is emitted, absorbed, or scattered by objects - particularly stars, planets and other celestial objects. By analysing the object’s light, astronomers can infer the physical properties of that object such as temperature, mass, luminosity and composition.
Spectroscopy can also include the study of the interaction between electrons, protons and ions.
To perform the measurements, a spectrometer, spectrophotometer or spectrograph is used.
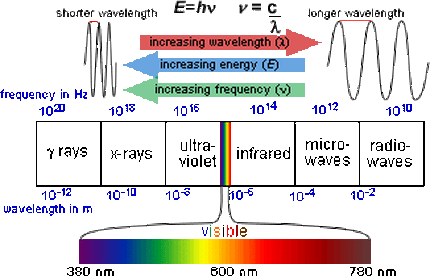
Spectroscopy involves the colour spectrum. This is abbreviated as "ROY G. BIV" (Red, Orange, Yellow, Green, Blue, Indigo, Violet). In addition to the colour spectrum, there are also gamma rays, x-rays, ultraviolet (UV) radiation, infrared (IR) radiation, microwaves and radio waves. This whole spectrum is referred to as the electromagnetic spectrum by physicists.
Today, spectroscopy is essentially a scientific measurement technique. It measures light that is emitted, absorbed, or scattered by objects - particularly stars, planets and other celestial objects. By analysing the object’s light, astronomers can infer the physical properties of that object such as temperature, mass, luminosity and composition.
Spectroscopy can also include the study of the interaction between electrons, protons and ions.
To perform the measurements, a spectrometer, spectrophotometer or spectrograph is used.
Spectroscopy involves the colour spectrum. This is abbreviated as "ROY G. BIV" (Red, Orange, Yellow, Green, Blue, Indigo, Violet). In addition to the colour spectrum, there are also gamma rays, x-rays, ultraviolet (UV) radiation, infrared (IR) radiation, microwaves and radio waves. This whole spectrum is referred to as the electromagnetic spectrum by physicists.
Physicists classify light waves by energies (wavelengths).
Spectroscopy has many different types. These include: absorption spectroscopy, fluorescence, x-ray, atomic emission, atomic absorption, atomic fluorescence, plasma emission, spark emission, visible absorption, ultraviolet, infrared, nuclear and photoemission.
Note that these are just some of the forms of spectroscopy, and not all of them, as there are many different analysis techniques, with a variety of approaches in investigating the absorbance, reflection and emission – just to name a few.
Spectroscopy has many different types. These include: absorption spectroscopy, fluorescence, x-ray, atomic emission, atomic absorption, atomic fluorescence, plasma emission, spark emission, visible absorption, ultraviolet, infrared, nuclear and photoemission.
Note that these are just some of the forms of spectroscopy, and not all of them, as there are many different analysis techniques, with a variety of approaches in investigating the absorbance, reflection and emission – just to name a few.